Subscriber Benefit
As a subscriber you can listen to articles at work, in the car, or while you work out. Subscribe NowThe race is on.
Around the world, more than 80 vaccine projects are under development by pharmaceutical companies and university research laboratories scrambling to find a way to slow or stop the novel coronavirus sweeping the globe.
At Purdue University, scientists are studying numerous molecules that inhibit the virus. At the Indiana University School of Medicine, researchers are exploring virus-like particles that might generate an immune response in humans.
At Eli Lilly and Co., hundreds of scientists, working in partnership with biotech companies in China and Canada, are examining antibodies taken from patients who have been infected with the hope of mass producing the most potent ones as a preventive treatment.
And at Butler University, researchers are focusing on an enzyme that cuts large viral proteins into smaller proteins, a process that accelerates the replication of the virus, to see if they can find a way to inhibit that process and help stop the spread.
It’s crunch time for researchers, as they try to find the best and fastest way to halt the rapid spread of a virus that has infected more than 5 million people and claimed more than 348,000 lives in just a few months, including more than 100,000 in the United States.
The stakes are gigantic: public health, the world economy and big potential profits for industry.
Fewer than 10 of the projects under development by pharmaceutical makers, biotech innovators, government researchers and academic laboratories have reached clinical trials—or the stage in drug development when researchers begin testing the vaccines in humans.
At least four or five possible vaccines “look pretty promising,” and one or two will be ready for large-scale testing by July, with others to follow soon thereafter, NIH Director Francis Collins told The Associated Press.
A mad dash
With political leaders pushing hard for progress, researchers are going full bore.

“There’s certainly enormous pressure on everybody in the scientific community and vaccine industry to come up with some kind of … vaccine or therapeutic to deal with those pathogens,” said Vidadi Yusibov, director of the Pharmaceutical Biotechnology Center at the Indiana Biosciences Research Institute, an independent not-for-profit in Indianapolis.
The pressure is coming from, among others, President Donald Trump, who said on May 15 that he wanted to make hundreds of millions of doses of a coronavirus vaccine available by the end of the year—something many scientists say is unrealistic.
Peter Jay Hotez, dean for the National School of Tropical Medicine at Baylor College of Medicine in Houston, told The Washington Post that, with some large trials likely to begin this summer, the most reasonable timeline for proving a vaccine safe and effective would be the second half of next year. He said even that would be “an absolute record.”
Some Indiana scientists agree.
“We typically see vaccines take 10 to 20 years from the moment they’re created in a laboratory to the moment they’re administered by a doctor at the bedside,” said Christopher Stobart, an assistant professor of biological sciences at Butler University, who has spent more than a decade studying viruses. “And so I think if you’re looking at timelines for the coronavirus … optimistically, 12 months is the fastest you can expect.”
The faster turnaround could happen by abbreviating some of the human testing, a process that often takes years—first in healthy volunteers, then in a small group of sick patients, and finally in huge groups of patients to get the best look at possible side effects across a wide population.
Researchers can possibly cut out the middle step, or speed up or shrink the size of one of the other steps.
No Indiana laboratory is known to be involved in a major program to develop a vaccine specifically for coronavirus. Several, however, are pursuing other therapies and treatments, such as Lilly’s quest to develop antibodies that can defend a cell from a pathogen by neutralizing its effect.
Lilly, which specializes in drugs for cancer and diabetes, has mostly shied away from vaccines. But it is developing something related, called “neutralizing antibodies.”
While vaccines work by providing “active immunity”—causing the body to produce antibodies to fight a virus—neutralizing antibodies provide a short-term or “passive immunity” by injecting antibodies from another person who has successfully fought the infection.

“It’s a real close cousin to vaccines,” said Andrew Adams, a Lilly scientist who is leading the Indianapolis-based drugmaker’s work in antibodies. “In the passive immunity efforts, we really just take the successful response from one person and then we magnify it and then put it into other people to either protect them or jump-start their immune response to the virus.”
Lilly competitors, meanwhile, are pushing hard to develop a vaccine. Britain-based pharmaceutical giant AstraZeneca is developing a vaccine at the University of Oxford and said it wants to manufacture tens of millions of doses by the end of this year.
Different approaches
Helping people’s bodies develop an immune response to a particular virus is a broad and complex field of medicine. There is no single type of vaccine; they fall into four main categories.
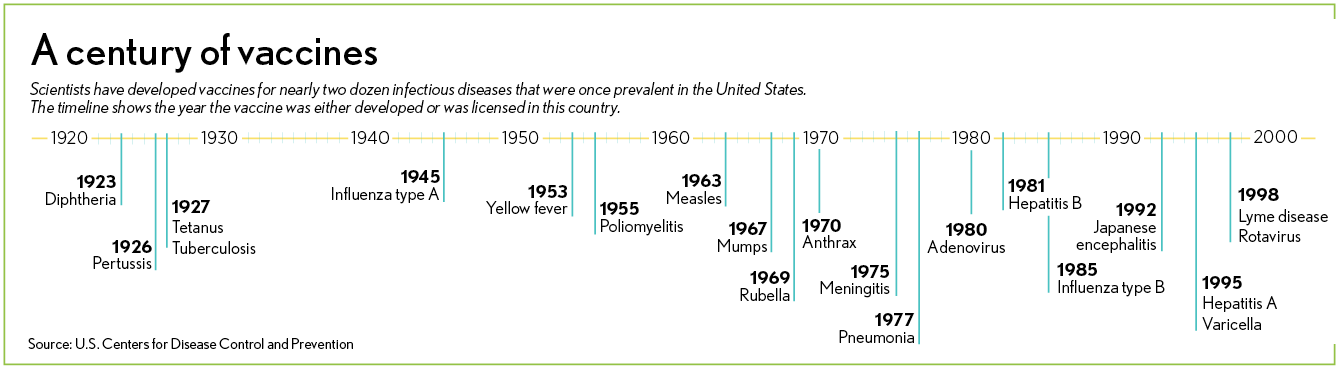
The first is live-attenuated vaccines, which use a weakened (attenuated) form of the germ that causes a disease. These vaccines are similar to the natural infection and can provide a strong, long-lasting immune response, sometimes for a lifetime. Examples include vaccines for measles, mumps, rubella, smallpox, chickenpox and yellow fever.

“And the idea would be that you will get vaccinated with that and you will be able to develop an immune response to that virus so that, when you’re challenged with the active, really infectious virus, that you would be able to fight it off,” said Mark H. Kaplan, chair of the Department of Microbiology and Immunology at the IU School of Medicine.
The second form is inactivated vaccines, which are the killed version of the germ that causes a disease. They generally don’t provide immunity as strong or for as long as the live vaccines. Examples include vaccines for hepatitis A, flu, polio and rabies.
The third form are what’s called subunit vaccines. As their name implies, they contain only part of a certain virus—such as its spikes or its protective sugar casing—which is used to trigger the body to fight the pathogen. This type of vaccine often gives a strong immune response. Examples include vaccines for hepatitis B, human papillomavirus, whooping cough, shingles, tetanus and pertussis.
“I have a feeling that the subunit vaccines are probably going to be the ones that are going to get to market the quickest,” Kaplan said. “They have the biggest potential for having the earliest impact because those are likely to have the best safety profile.”
The last and newest type are nucleic-acid vaccines. With this type, a person is injected with genetically engineered DNA or RNA of the virus that causes the body’s cells to make viral proteins to produce an immunological response. No DNA or RNA vaccines have been approved yet for human use.
Frightening speed
It’s still unknown which approach will be most suitable for fighting the coronavirus. So researchers are looking at as many as possible. It might take two or three or four vaccines to find one that is effective.

“We don’t know whether the first few vaccines are even going to work,” said Richard J. Kuhn, a professor of biological sciences in Purdue University’s Institute of Inflammation, Immunology and Infectious Disease. “The hope is they’re going to work, but I wouldn’t say that we are 100% confident that any particular vaccine is going to work right now.”
In his laboratory, Kuhn said, he is looking at “a number of molecules that inhibit the virus,” although he is not actively working on a COVID-19 vaccine.
Viruses, which cause disease by invading cells, are extremely diverse and plentiful. Some scientists estimate Earth is home to hundreds of thousands of viruses, although most are relatively harmless and localized. But the novel coronavirus—formally known as SARS-CoV-2, is the most dangerous pandemic in generations.
COVID-19 is a respiratory illness caused by the large family of viruses called coronavirus, which have included SARS and MERS.
The original SARS (severe acute respiratory syndrome) broke out in 2004 and caused just 774 deaths before it was contained. It was more aggressive and lethal than SARS-CoV-2.
But scientists say SARS-CoV-2 spreads at frightening speed, sometimes with hidden symptoms. That allows each infected person to infect several others. Sometimes, symptoms don’t appear for two weeks, and many people don’t get any symptoms at all. That makes it tougher to hold back than the original SARS, which was easier to trace and contain.
It also raises the ante significantly for a vaccine or other treatment, whereas the original SARS stopped spreading before a vaccine was ever developed. Since 2004, no known cases of the original SARS have been reported.
 Modes of transmission
Modes of transmission
How a virus spreads can determine how many lives it is likely to claim. The Ebola virus, which has been around for decades, is spread by direct contact with body fluids, such as blood from infected humans or animals. But relatively few people have widespread exposure to body fluids, so the disease has been much smaller. The largest outbreak was in West Africa from 2013 to 2106, which claimed 11,323 lives.
“Now SARS, on the other hand, is an incredibly contagious virus,” said Yusibov at the IBRI in Indianapolis. “And it’s spread by respiratory routes by air droplets, by air association, and it’s a lot more dangerous from that sense. And that’s why, in a very short period of time, it was all over the globe.”
Other viruses come and go without much notice. In recent years, for example, Massachusetts, New York, Rhode Island and other states saw an outbreak of a virus called eastern equine encephalitis. Fewer than 20 people died.
“It was small, but it was still a bit problematic,” said Kuhn at Purdue. “So we were involved in developing vaccines and therapeutics for that.”
At IU, several researchers are looking at different approaches for the latest coronavirus.
Kaplan said he is working with Yuichiro Takagi, associate professor of biochemistry and molecular biology at the medical school, to research ways to develop a virus-like particle that resembles a hybrid between the virus and the subunit vaccine.
“It essentially allows you to generate virus-like particles that have multiple proteins,” Kaplan said. “So you can generate an immune response against those, but it doesn’t have anything infectious and so you don’t have the danger of getting an infection from inactivated vaccines.”
In Bloomington, IU Associate Professor of Biology John Patton is trying to develop a vaccine that will protect children against COVID-19. He is using years of experience working on vaccines for rotavirus, an agent that commonly causes diarrhea and vomiting in infants and young children.
“We realized that we can reengineer rotavirus to make virus strains that produce the coronavirus spike protein,” Patton said in an IU news release. “So when a child is immunized with rotavirus vaccine, they not only will mount a protective immune response against rotavirus, but in addition will likely have a similar protective immune response to COVID-19.”
Resolving the crisis
Overall, scientists say ending the COVID-19 crisis has three main avenues.
The first option—the worst-case scenario—is for enough people on the planet to get infected that the human race develops a widespread immunity, sometimes called herd immunity. The virus is unable to spread efficiently because so many people’s immune systems are able to fight it off. But that would likely involve millions of deaths.
The second option—the best-case scenario—is developing a vaccine. The result would be far fewer illnesses and deaths.
“And in that scenario, you’d still need to vaccinate, probably at least half if not two-thirds of the planet, to start getting to that herd immunity, that level of protection where the virus can’t spread well,” Stobart at Butler University said.
The third option is to develop an effective drug treatment, so that, if people get infected and go to the hospital, they can be treated and recover.
So thousands of researchers are working on some variation of the last two options.
“It’s likely,” Stobart said, “that it will be a combination of the two that finally stops the virus in its tracks.”•
Please enable JavaScript to view this content.


Editor's note: You can comment on IBJ stories by signing in to your IBJ account. If you have not registered, please sign up for a free account now. Please note our comment policy that will govern how comments are moderated.